Matter and Materials: Identify and apply knowledge of organic molecules in everyday life and industrial context
Unit 4: Applications of organic molecules
Emma Harrage
Unit outcomes
By the end of this unit you will be able to:
- Understand that crude oil is distilled into different components.
- Understand the process of fractional distillation and the uses of the different compounds separated from crude oil.
What you should know
Before you start this unit, make sure you can:
- Understand hydrocarbons and functional groups. Refer to level 4 subject outcome 5.3 unit 1 if you need help with this.
- Recall separation techniques. Refer to level 2 subject outcome 6.2 unit 2 if you need help with this.
Introduction
Parts of the text in this unit were sourced from Siyavula Physical Science Gr 12 Learner’s Book, Chapter 4, released under a CC-BY licence.
In this unit you will learn about the formation of crude oil and how it is separated by fractional distillation into useful components.
Formation of crude oil
Fossil fuels are non-renewable sources of energy formed from the organic matter of plants and microorganisms that lived millions of years ago. This energy was originally captured via photosynthesis by living organisms such as plants, algae, and photosynthetic bacteria.
Fossil fuels are non-renewable because their formation took millions of years. Furthermore, higher productivity in the ancient environment allowed for more fossil fuel accumulation, meaning that the fossil fuel reserves available now could not necessarily be regenerated millions of years in the future.
Fossil fuels are composed primarily of hydrocarbons (molecules of just carbon and hydrogen), but they contain lesser amounts nitrogen, sulfur, oxygen, and other elements as well. Both oil and natural gas are fossil fuels found underground that formed from marine microorganisms. Oil (petroleum) is a liquid fossil fuel and consists of a variety of hydrocarbons while natural gas is a gaseous fossil fuel that consists of mostly methane and other small hydrocarbons.
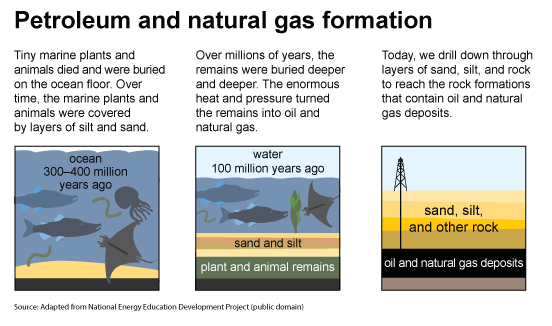
Oil and natural gas formed from ancient marine microorganism (plankton). When plankton died, they were buried in sediments. As with coal, oxygen-poor conditions limited decomposition. As sediments continued to accumulate, the dead organisms were further buried. High temperature and pressure over millions of years ultimately produced oil and natural gas from these dead organisms.
Alkanes as fossil fuels
Crude oil when extracted is a thick black liquid. It is a complex mixture of different chemical compounds known as hydrocarbons ranging from methane to asphalt. The composition of crude oil varies with the type of crude oil and how it is extracted. Hydrocarbons such as alkanes are the main fraction of crude oil. It may also contain aromatic compounds and other compounds with nitrogen, and sulfur along with metals such as copper, nickel, vanadium, and iron. Hence crude oil is the main source of these organic compounds and fractional distillation is carried out to make these organic compounds available and usable.
Organic molecules vary in their physical and chemical properties due to their different carbon chain lengths and molecular masses. The melting point, boiling point, viscosity and colour are all related to the molecular mass and structure of the molecule. The boiling point of organic compounds increases with increasing molecular mass.
Fractional distillation
As mentioned, crude oil contains a wide range of organic compounds that differ in the length of their carbon chains and boiling points. is the process to separate these individual fractions from each other. The whole process is based on the principle that different substances boil at different temperatures. For example, crude oil contains kerosene and naphtha, both are useful fractions but have different usage and applications. Naphtha can be used to form petrol for cars while kerosene is used for jet fuel. Both have different carbon chain lengths and structures. During distillation, the mixture of kerosene and naphtha is first vaporised then they are cooled down; the kerosene condenses at a higher temperature than the naphtha. As the mixture cools, the kerosene condenses first, and the naphtha condenses later. This is how fractional distillation works. The whole process can be divided into steps.
- The pre-treatment step: The crude oil extracted from an oil well could be an emulsion containing some rock, salt and water. In the first step, crude oil is pre-treated to remove this water and salt (sodium chloride or brine). The desalting step is important because the presence of sodium chloride can generate highly corrosive hydrogen chloride gas during the distillation process. This hydrogen chloride, being a strong acid may cause severe damage to the processing equipment. The desalting can be carried out by heating the crude oil under pressure or adding demulsifying agents e.g. soap, fatty acids, and surfactants to break the emulsion.
- Distillation: The crude oil is heated to [latex]\scriptsize \displaystyle 700\text{ }{}^\circ \text{C}[/latex] using heat exchangers and is converted to vapours. These vapours are sent to a distillation tower. The whole process is typically conducted in a vertical tower or column.
.
The tower has a temperature gradient, which means it is hot at the bottom and cooler at the top. It contains a series of trays installed to collect fractions that are finally removed via outlets. Because different fractions have different boiling points, they condense at different heights at a different level of the column. The large molecules with higher molecular mass stay in the bottom while smaller molecules with low molecular mass travel upward and condense at their respective collection tray. The solid waxy residue is removed from the bottom while molecules with very low molecular mass stay as gases and are collected from the top. They are called petroleum gas.
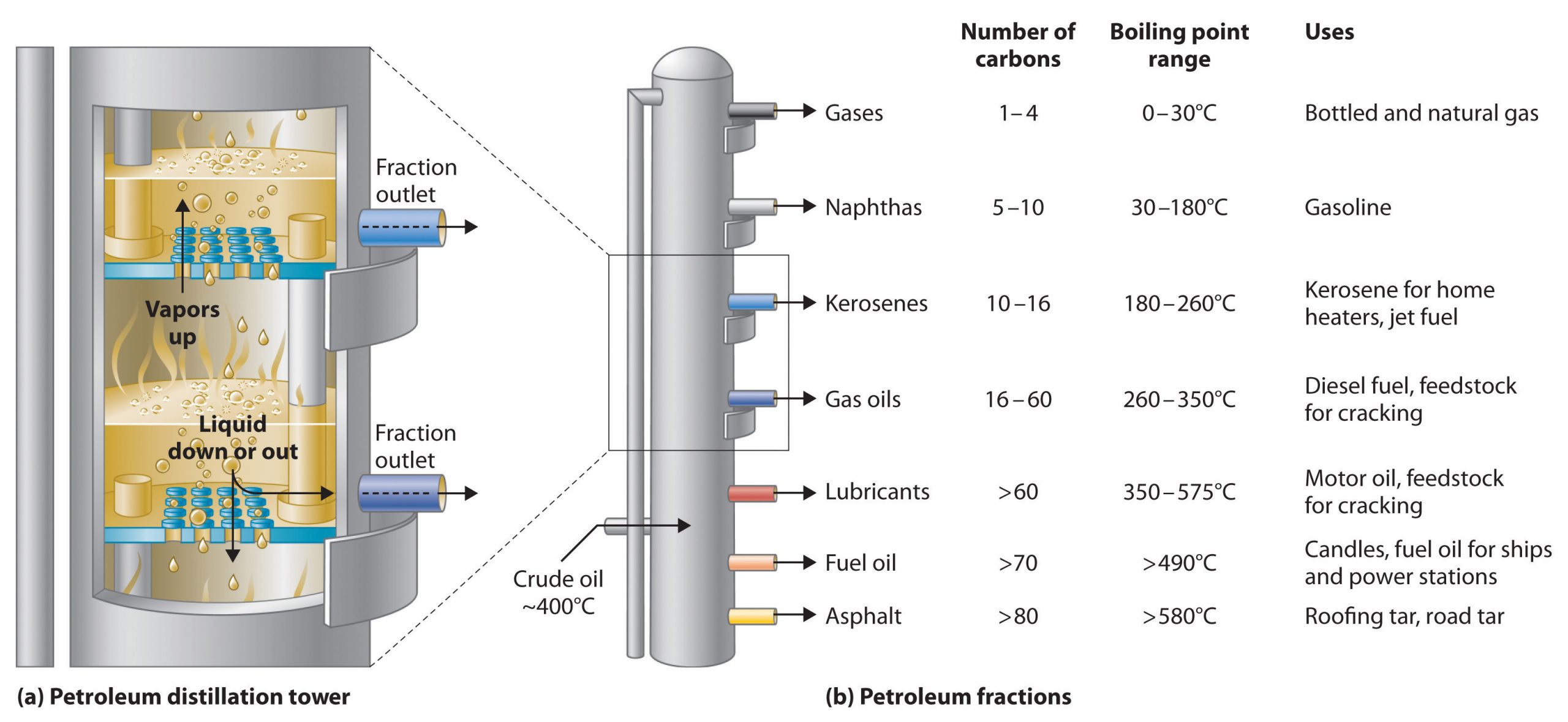
In figure 2, in diagram (a) you can see a distillation column used for separating petroleum fractions. In diagram (b) the petroleum fractions condense at different temperatures, depending on the number of carbon atoms in the molecules, and are drawn off from the column. The most volatile components (those with the lowest boiling points) condense at the top of the column, and the least volatile (those with the highest boiling points) condense at the bottom.
is the process of breaking carbon-carbon bonds in long-chain hydrocarbons to form simpler, shorter-chain hydrocarbons.
There is a high demand for shorter alkanes but they are in short supply. Long chain hydrocarbons can be broken down into smaller chain alkanes and alkenes by cracking. Cracking is a thermal decomposition reaction. The hydrocarbon is vapourised and passed over a hot catalyst and this splits the long chains apart.
Hydrocarbon cracking is an important industrial process. These compounds include shorter alkanes and alkenes. A few examples of cracking hydrocarbons are given in figure 3.
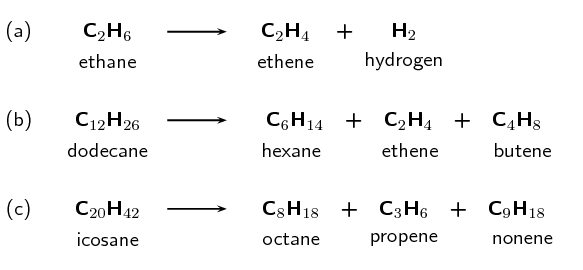
It is possible to separate the products of hydrocarbon cracking and obtain specific products from a crude oil mix through fractional distillation. When the crude oil evaporates the gas bubbles through a tray that is kept at a certain temperature. The alkanes and alkenes that condense at that temperature will then condense in the tray. For example, if the tray is kept at [latex]\scriptsize \displaystyle 170\text{ }{}^\circ \text{C}[/latex] the product will be paraffin oil.
A fractionating column has a series of these trays (see figure 2), each at a constant temperature. This means that many compounds can be separated from the crude oil mix.
Bitumen for tar roads is collected at the bottom of the fractionating column. These are all compounds with more than [latex]\scriptsize \displaystyle 70[/latex] carbon atoms. The temperature decreases as you move up the column. As the gases rise, compounds with different length carbon chains condense until only the chains with [latex]\scriptsize \displaystyle 1-4[/latex] carbon atoms are collected at the top of the column. These are used for liquid petroleum gas.
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{3}}}..{{\text{C}}_{\text{4}}}[/latex] | Propane and butane can be liquefied at low pressures, and are used, for example, in a propane gas burner, or as propellants in aerosol sprays. Butane is used in cigarette lighters. |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{5}}}..{{\text{C}}_{\text{8}}}[/latex] | The alkanes from pentane to octane are highly volatile liquids and good solvents for nonpolar substances. They are used as fuels in internal combustion engines. |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{9}}}..{{\text{C}}_{{\text{16}}}}[/latex] | Alkanes from nonane to hexadecane are liquids of higher viscosity, being used in diesel and aviation fuel (kerosene). The higher melting points of these alkanes can cause problems at low temperatures and in polar regions, where the fuel becomes too viscous. |
| [latex]\scriptsize \displaystyle {{\text{C}}_{{\text{17}}}}..{{\text{C}}_{{\text{35}}}}[/latex] | Alkanes with [latex]\scriptsize \displaystyle 17[/latex] to [latex]\scriptsize \displaystyle 35[/latex] carbon atoms form the major components of lubricating oil. They also act as anti-corrosive agents, as their hydrophobic nature protects the metal surface from contact with water. Solid alkanes are also used as paraffin wax in candles. |
| [latex]\scriptsize \displaystyle \gt {{\text{C}}_{{35}}}[/latex] | Alkanes with a chain length above [latex]\scriptsize \displaystyle 35[/latex] carbon atoms are found in bitumen (used in road surfacing). These higher alkanes have little chemical and commercial value and are usually split into lower alkanes by cracking. |
Note
To consolidate your understanding of fractional distillation you can watch this video called Fractional Distillation by Fuse Schools.
Combustion of alkanes
Alkanes are our most important fossil fuels. The (burning) of alkanes is highly exothermic.
Methane is used as a fuel for domestic heating, cooking, and heating water. Methane is important for electricity generation by burning it as a fuel in a gas turbine or steam generator. Compared to other hydrocarbon fuels, methane produces less carbon dioxide for each unit of heat released. In many cities, methane is piped into homes for domestic heating and cooking.
In the complete combustion reaction of alkanes, carbon dioxide and water are released along with energy. Fossil fuels are burnt for the energy they release. The general reaction for the combustion of an alkane as a fossil fuel is:
alkane + oxygen → carbon dioxide + water + energy
For example, the combustion of propane:
[latex]\scriptsize {{\text{C}}_{\text{3}}}{{\text{H}}_{{8\text{(g)}}}}\text{+ 5}{{\text{O}}_{\text{2}}}_{{\text{(g)}}}\to 3\text{C}{{\text{O}}_{{\text{2(g)}}}}+\text{4}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\text{(g)}}}}+\text{ energy}[/latex]
Propane is a flammable gas used in blowtorches for soldering.
Example 4.1
Balance the following equation: [latex]\scriptsize \displaystyle {{\text{C}}_{\text{4}}}{{\text{H}}_{{\text{10}\left( \text{g} \right)}}}\text{+}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{ }\to \text{ C}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{+}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{g} \right)}}}[/latex].
Solution
Step 1: Balance the carbon atoms
There are [latex]\scriptsize \displaystyle 4[/latex] carbon atoms on the left. There is [latex]\scriptsize \displaystyle 1[/latex] carbon atom on the right. Add a [latex]\scriptsize \displaystyle 4[/latex] in front of the [latex]\scriptsize \displaystyle \text{C}{{\text{O}}_{\text{2}}}[/latex] molecule on the right: [latex]\scriptsize \displaystyle {{\text{C}}_{\text{4}}}{{\text{H}}_{{\text{10}\left( \text{g} \right)}}}\text{+}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{ }\to \text{ 4C}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{+}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{g} \right)}}}[/latex]
Step 2: Balance the hydrogen atoms
There are [latex]\scriptsize \displaystyle 10[/latex] hydrogen atoms on the left. There are [latex]\scriptsize \displaystyle 2[/latex] hydrogen atoms on the right. Add a [latex]\scriptsize \displaystyle 5[/latex] in front of the [latex]\scriptsize \displaystyle {{\text{H}}_{\text{2}}}\text{O}[/latex] molecule on the right: [latex]\scriptsize \displaystyle {{\text{C}}_{\text{4}}}{{\text{H}}_{{\text{10}\left( \text{g} \right)}}}\text{+}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{ }\to \text{ 4C}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{+ 5}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{g} \right)}}}[/latex]
Step 3: Balance the oxygen atoms
There are [latex]\scriptsize \displaystyle 2[/latex] oxygen atoms on the left. There are [latex]\scriptsize \displaystyle 13[/latex] oxygen atoms on the right. Divide the number of O atoms on the right by [latex]\scriptsize 2[/latex] to get [latex]\scriptsize \displaystyle \frac{{13}}{2}[/latex], this is the number of [latex]\scriptsize \displaystyle {{\text{O}}_{\text{2}}}[/latex] molecules required on the left:
[latex]\scriptsize \displaystyle {{\text{C}}_{\text{4}}}{{\text{H}}_{{\text{10}\left( \text{g} \right)}}}\text{+ }\displaystyle \frac{{13}}{2}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{ }\to \text{ 4C}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{+ 5}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{g} \right)}}}[/latex]
This is acceptable but it is better for all numbers to be whole numbers.
Step 4: Make sure all numbers are whole numbers
There is [latex]\scriptsize \displaystyle \frac{{13}}{2}[/latex] in front of the [latex]\scriptsize \displaystyle {{\text{O}}_{\text{2}}}[/latex] while all other numbers are whole numbers. So multiply the entire equation by[latex]\scriptsize 2[/latex]:
[latex]\scriptsize \displaystyle \text{2}{{\text{C}}_{\text{4}}}{{\text{H}}_{{\text{10}\left( \text{g} \right)}}}\text{+ 13}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{ }\to \text{ 8C}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{+ 10}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{g} \right)}}}[/latex]
The combustion of hydrocarbons is an exothermic process otherwise there would not be much point in burning them to produce energy for fuel and heat.
Gasoline, which distils at temperatures between [latex]\scriptsize 40\text{ - 205 }{}^\circ \text{C}[/latex] is used as fuel for internal combustion engines. Gas oil, which distils at temperatures between [latex]\scriptsize 275-400\text{ }{}^\circ \text{C}[/latex] is used in diesel engines and industrial furnaces.
Summary
In this unit you have learnt the following:
- Crude oil can be separated by fractional distillation into simpler hydrocarbons.
- Simple hydrocarbons can be used as fuels
- Alkanes, including methane, are important as fuels because of the amount of heat they produce during combustion.
Unit 4: Assessment
Suggested time to complete: 15 minutes
- Describe the conditions required for the formation of crude oil.
- Briefly describe the process of fractional distillation.
- Explain the relationship between the number of carbon atoms and the boiling point of the hydrocarbon.
- Is the combustion of alkanes exothermic or endothermic? What does that mean?
- Balance the following complete combustion equation:
[latex]\scriptsize \displaystyle ~{{\text{C}}_{\text{7}}}{{\text{H}}_{{\text{16}\left( \text{l} \right)}}}\text{+}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{ }\to \text{ C}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{+}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{g} \right)}}}[/latex] - Petrol is in fact not pure [latex]\scriptsize \displaystyle {{\text{C}}_{\text{8}}}{{\text{H}}_{{\text{18}}}}[/latex] but a mixture of various alkanes. The octane rating of petrol refers to the percentage of the petrol which is [latex]\scriptsize \displaystyle {{\text{C}}_{\text{8}}}{{\text{H}}_{{\text{18}}}}[/latex]. For example, [latex]\scriptsize \displaystyle 93[/latex] octane fuel contains [latex]\scriptsize \displaystyle 93\%\text{ }{{\text{C}}_{\text{8}}}{{\text{H}}_{{\text{18}}}}[/latex] and [latex]\scriptsize \displaystyle 7\%[/latex] other alkanes.
Write a balanced equation for the chemical reaction which takes place when [latex]\scriptsize \displaystyle {{\text{C}}_{\text{8}}}{{\text{H}}_{{\text{18}}}}[/latex] burns in oxygen.
The full solutions are at the end of the unit.
Unit 4: Solutions
Unit 4: Assessment
- Intense heat and pressure from hundreds of layers of dead plants and animals being compressed together over millions of years.
- Heating the crude oil mixture to different temperature ranges causes different fractions to vaporise. These vapours can be collected by cooling and condensing them in a separate receiving tube.
- The higher the number of carbon atoms, the higher the boiling point. This is because more carbon atoms means larger molecules and stronger attractions between molecules, needing more energy to overcome the forces between them.
- It is exothermic. This means that energy is released during the combustion of an alkane.
- [latex]\scriptsize \displaystyle {{\text{C}}_{\text{7}}}{{\text{H}}_{{\text{16}\left( \text{l} \right)}}}\text{+11}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{ }\to \text{ 7C}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{+ 8}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{g} \right)}}}[/latex]
- [latex]\scriptsize \displaystyle \text{2}{{\text{C}}_{\text{8}}}{{\text{H}}_{{\text{18}\left( \text{l} \right)}}}\text{+25}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{ }\to \text{ 16C}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{+18}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{g} \right)}}}[/latex]
Media Attributions
- Fig 1 © Libre text is licensed under a CC BY-NC-SA (Attribution NonCommercial ShareAlike) license
- Fig 2 © Libre text is licensed under a CC BY-NC-SA (Attribution NonCommercial ShareAlike) license
- Fig 3 © Siyavula is licensed under a CC BY-NC (Attribution NonCommercial) license
the separation of a liquid mixture into fractions of differing in boiling points and hence chemical composition by means of distillation
breaking carbon-carbon bonds in long-chain hydrocarbons to form simpler, shorter-chain hydrocarbons
burning a substance; the substance reacts with an oxidising agent (e.g. oxygen), and heat and light are released
