Matter and Materials: Identify and apply knowledge of organic molecules in everyday life and industrial context
Unit 3: Physical Properties of Organic molecules
Emma Harrage
Unit outcomes
By the end of this unit you will be able to:
- Understand physical and chemical properties of organic molecules.
What you should know
Before you start this unit, make sure you:
- Understand hydrocarbons and functional groups. Refer to level 4 subject outcome 5.3 unit 1 if you need help with this.
- Can name organic molecules. Refer to level 4 subject outcome 5.3 unit 2 if you need help with this.
- Understand polarity. Refer to level 3 subject outcome 5.2 unit 5 if you need help with this.
- Can recall the different types of bonding in covalent molecules. Refer to level 3 subject outcome 5.3 unit 1 if you need help with this.
Introduction
Parts of the text in this unit were sourced from Siyavula Physical Science Gr 12 Learner’s Book, Chapter 4, released under a CC-BY licence.
In this unit you will learn about the chemical and physical properties of hydrocarbons. This will include density, melting and boiling points, and how chain length affects these properties.
Physical properties
The types of intermolecular forces that occur in a substance will affect its physical properties, such as its phase, melting point, and boiling point. You should remember from the kinetic theory of matter that the phase of a substance is determined by how strong the forces are between its particles. The weaker the forces, the more likely the substance is to exist as a gas. This is because the particles can move far apart since they are not held together very strongly. If the forces are very strong, the particles are held closely together in a solid structure. Remember also that the temperature of a material affects the energy of its particles. The more energy the particles have, the more likely they are to be able to overcome the forces that are holding them together. This can cause a change in phase.
The physical properties of any compound depend largely on:
- its mass
- its type of intermolecular and intramolecular forces of attraction
- its melting point and boiling point.
Compounds that contain very similar atoms can have very different properties depending on how those atoms are arranged. This is especially true when they have different functional groups. Table 1 shows some properties of different homologous series.
| Homologous series | Typical smell |
Example | Formula | Melting point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) | Boiling point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) | Phase (at [latex]\scriptsize \displaystyle 25\text{ }{}^\circ \text{C}[/latex]) |
| alkane | odourless | ethane | [latex]\scriptsize {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}[/latex] | [latex]\scriptsize -183[/latex] | [latex]\scriptsize -89[/latex] | gas |
| alkene | sweet/musky | ethene | [latex]\scriptsize {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}[/latex] | [latex]\scriptsize -169.2[/latex] | [latex]\scriptsize -103.7[/latex] | gas |
| haloalkane | almost odourless | chloro ethane | [latex]\scriptsize {{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}\text{Cl}}[/latex] | [latex]\scriptsize -139[/latex] | [latex]\scriptsize 12.3[/latex] | gas |
| alcohol | sharp | ethanol | [latex]\scriptsize {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}\text{O}[/latex] | [latex]\scriptsize -114[/latex] | [latex]\scriptsize 78.4[/latex] | liquid |
| alkyne | odourless | ethyne | [latex]\scriptsize \text{C}_2 \text{H}_2[/latex] | [latex]\scriptsize -80.8[/latex] | [latex]\scriptsize -84[/latex] | gas |
Listed in the table are the common smells and other physical properties for common homologous series. Only one representative example from each homologous series is provided. This does not mean that all compounds in that series have the same properties. For example, short-chain and long-chain alkanes are generally odourless, while those with moderate chain length (approximately [latex]\scriptsize \displaystyle 6-12[/latex] carbon atoms) smell like petrol.
Intermolecular forces
Intermolecular forces affect the boiling and melting points of substances. Substances with weak intermolecular forces will have low melting and boiling points as less energy (heat) is needed to overcome these forces. Those with strong intermolecular forces will have high melting and boiling points as more energy (heat) is required to overcome these forces. When the temperature of a substance is raised beyond its melting or boiling point the intermolecular forces are not weakened. Rather, the molecules have enough energy to overcome those forces.
| Name | Main Intermolecular forces | Molecular mass ([latex]\scriptsize \text{g}\text{.mo}{{\text{l}}^{{\text{-1}}}}[/latex]) |
Melting point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) | Boiling point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) | Phase (at ) |
| ethane | induced-dipole | [latex]\scriptsize \displaystyle 30.06[/latex] | [latex]\scriptsize \displaystyle -183[/latex] | [latex]\scriptsize \displaystyle -89[/latex] | gas |
| dimethyl ether | dipole-dipole | [latex]\scriptsize \displaystyle 46.06[/latex] | [latex]\scriptsize \displaystyle -141[/latex] | [latex]\scriptsize \displaystyle -24[/latex] | gas |
| chloroethane | dipole-dipole | [latex]\scriptsize \displaystyle 64.5[/latex] | [latex]\scriptsize \displaystyle -139[/latex] | [latex]\scriptsize \displaystyle 12.3[/latex] | gas |
| pentane | induced-dipole | [latex]\scriptsize \displaystyle 72.12[/latex] | [latex]\scriptsize \displaystyle -130[/latex] | [latex]\scriptsize \displaystyle 36[/latex] | liquid |
| propan-1-ol | hydrogen bonds | [latex]\scriptsize \displaystyle 60.08[/latex] | [latex]\scriptsize \displaystyle -126[/latex] | [latex]\scriptsize \displaystyle 97[/latex] | liquid |
| ethanol | hydrogen bonds | [latex]\scriptsize \displaystyle 46.06[/latex] | [latex]\scriptsize \displaystyle -114[/latex] | [latex]\scriptsize \displaystyle 78.4[/latex] | liquid |
| butan-1-ol | hydrogen bonds | [latex]\scriptsize \displaystyle 74.1[/latex] | [latex]\scriptsize \displaystyle -90[/latex] | [latex]\scriptsize \displaystyle 118[/latex] | liquid |
As the strength of the intermolecular forces increases (from top to bottom in table 2) the melting and boiling points increase. The stronger the intermolecular forces the more likely a substance is to be a liquid or a solid at room temperature.
Flammability
Flammability is a measure of how easy it would be for a substance to catch alight and burn. The flash point of a substance is the lowest temperature that is likely to form a gaseous mixture you could set alight. If a liquid has a low enough flash point it is considered flammable (able to be ignited easily) while those with higher flash points are considered non-flammable. A substance that is classified as non-flammable can still be forced to burn, but it will not ignite easily.
| Name | Main intermolecular forces | Vapour pressure (kPa at [latex]\scriptsize 20\text{ }{}^\circ \text{C}[/latex]) | Flash point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) | Flammability |
| ethane | induced-dipole | [latex]\scriptsize \displaystyle 3~750[/latex] | [latex]\scriptsize \displaystyle -135[/latex] | very high |
| propane | induced-dipole | [latex]\scriptsize \displaystyle 843[/latex] | [latex]\scriptsize \displaystyle -104[/latex] | very high |
| dimethyl ether | dipole-dipole | [latex]\scriptsize \displaystyle 510[/latex] | [latex]\scriptsize \displaystyle 41[/latex] | very high |
| butane | induced-dipole | [latex]\scriptsize \displaystyle 204[/latex] | [latex]\scriptsize \displaystyle -60[/latex] | very high |
| chloroethane | dipole-dipole | [latex]\scriptsize \displaystyle 132.4[/latex] | [latex]\scriptsize \displaystyle -50[/latex] | very high |
| pentane | induced-dipole | [latex]\scriptsize \displaystyle 57.9[/latex] | [latex]\scriptsize \displaystyle -49[/latex] | very high |
| propanone | dipole-dipole | [latex]\scriptsize \displaystyle 24.6[/latex] | [latex]\scriptsize \displaystyle -17[/latex] | high |
| ethanol | hydrogen bonds | [latex]\scriptsize 5.8[/latex] | [latex]\scriptsize 17[/latex] | high |
| water | hydrogen bonds | [latex]\scriptsize 2.3[/latex] | – | very low |
| propan-1-ol | hydrogen bonds | [latex]\scriptsize 2[/latex] | [latex]\scriptsize 22[/latex] | high |
| butan-1-ol | hydrogen bonds | [latex]\scriptsize 0.6[/latex] | [latex]\scriptsize 35[/latex] | high |
When a substance is in a liquid or solid state there will be some molecules in the gas state. These molecules have enough energy to overcome the intermolecular forces holding most of the substance in the liquid or solid phase. These gas molecules exert a pressure on the liquid or solid (and the container) and that pressure is the of that compound. The weaker the intermolecular forces within a substance the higher the vapour pressure will be. Compounds with higher vapour pressures have lower flash points and are therefore more flammable.
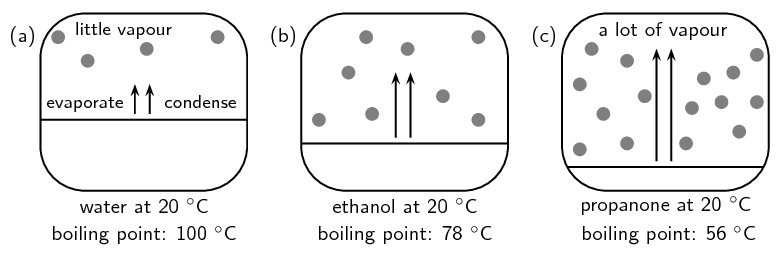
Exercise 3.1
Use the information in the table below to answer the following questions.
| Name | Boiling point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) | |
| A | Ethyne | [latex]\scriptsize -84.0[/latex] |
| B | Ethyne | [latex]\scriptsize -89.0[/latex] |
| C | Ethanol | [latex]\scriptsize 78.4[/latex] |
- Which of the compounds listed in the table are gases at room temperature?
- Name the main type of intermolecular forces for A, B and C.
- Briefly explain the difference in boiling point between B and C.
- Draw the structural representations of A, B and C.
The full solutions are at the end of the unit.
The physical properties of alkanes
Alkanes are the simplest type of hydrocarbons. There are only [latex]\scriptsize \displaystyle \text{C-H}[/latex] bonds throughout the whole structure of alkanes. Alkanes do not have any functional group. As the number of carbon atoms increases, the size of the molecule also increases.
As there are a greater number of atoms, there are a greater number of electrons, which means stronger intermolecular forces. So as the number of carbon atoms in alkanes increase, their melting and boiling points also increases. Alkanes with low molecular mass such as methane, ethane and propane are gas at room temperature, alkanes with greater molecular mass such as octane are liquid at room temperatures.
[latex]\scriptsize \displaystyle \text{C-H}[/latex] bonds are non-polar, which means there are only weak London forces between molecules. So most alkanes have a low melting and boiling points. Most alkanes are usually , they easily evaporate.
Alkanes are completely insoluble in water, because in water there is stronger hydrogen bonding.
The physical properties of alkenes
Alkenes have a carbon-to-carbon double bond in their structure ([latex]\scriptsize \displaystyle \text{C=C}[/latex]). This gives them a unique chemical property and similar trends in physical properties. There are also [latex]\scriptsize \displaystyle \text{C-H}[/latex] bonds throughout their structure. Both [latex]\scriptsize \displaystyle \text{C=C}[/latex] and [latex]\scriptsize \displaystyle \text{C-H}[/latex] bonds are non-polar, which means there are no strong dipole-dipole interactions present between molecules. There are only weak London forces. Therefore alkenes have low melting and boiling points. Most of the short chain alkenes are usually gases at room temperature.
Again, as the carbon number increases, the molecule gets bigger, which means more electrons and stronger intermolecular forces between the molecules. So as the number of carbon atoms in the chain increases, the melting and boiling points of the alkenes also increases.
Alkenes are also insoluble in water. Like alkanes, there are only weak London forces between the alkene molecules, while water molecules have a much stronger hydrogen bonding between them. Hence, they do not mix.
The physical properties of alkynes
The properties of alkynes pretty much follow the same pattern of those of alkanes and alkenes. Alkynes are unsaturated hydrocarbons that have a [latex]\scriptsize \text{C}\equiv \text{C}[/latex]. All alkynes are odourless and colourless except for ethylene which has a slight distinctive odour.
The first three alkynes are gases, and the next eight are liquids. All alkynes with longer chains than these eleven are solids.
The boiling point and melting point of alkynes increases as their molecular mass increases. The boiling points of alkynes are slightly higher than those of their corresponding alkenes, because of one extra bond at the carbon site.
The physical properties of alcohols
Alcohols have an [latex]\scriptsize \displaystyle \text{-OH}[/latex] group present in their structure, which is highly polar, which makes the alcohol molecules polar too. Alcohols are so polar, that they form very strong hydrogen bonds between molecules which are much stronger than London forces. Therefore alcohols have much higher melting and boiling points when compared to both alkanes and alkenes. Most alcohols are usually liquid at room temperature.
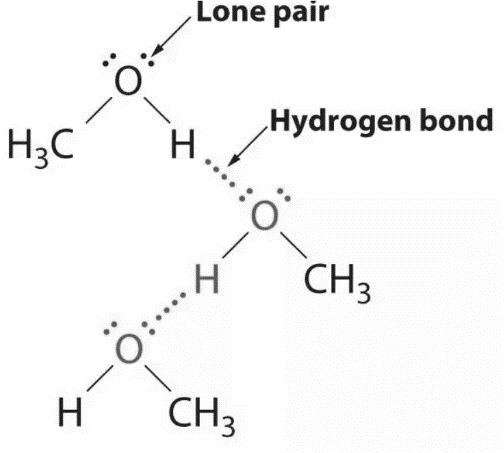
Alcohols are highly soluble in water, because both water and alcohols can form hydrogen bonds with each other.
The physical properties of haloalkanes
As the chain length increases in the homologous series of the haloalkanes group, differences can be seen because of the difference in atomic mass of the compounds.
There is a large electronegativity difference between a halogen atom and a carbon atom, resulting in highly polarised molecules. The higher molecular mass and greater polarity as compared to the parent hydrocarbon molecule results in stronger intermolecular forces of attraction: dipole-dipole and van der Waals. Boiling points depend upon the intermolecular forces of attraction and hence the boiling points of chloro-alkanes, bromo-alkanes and iodo-alkanes are considerably higher than those of the hydrocarbons of comparable molecular mass.
As the chain length increases in the homologous series of haloalkanes, the forces of attraction become stronger due to the increase in molecular size and mass, hence the boiling point increases. But, as with all hydrocarbons, the boiling point decreases with branching.
chloromethane, bromomethane, chloroethane and some chlorofluoromethanes are gases at room temperature. Higher members are liquids or solids.
The melting point of a compound depends upon the strength of the intermolecular forces between the molecules. The melting point also follows the same trend as the boiling point.
Haloalkanes are slightly soluble in water. This is because of the relatively larger amount of energy required to break the bond between halogen and carbon and the smaller amount of energy released when the bond is formed after the dissolution of the ion and water. The more halogens that are substituted, the less volatile the haloalkane becomes. Take for example the haloalkane series shown in figure 3.
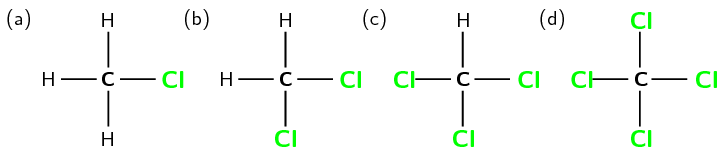
Physical properties and chain length
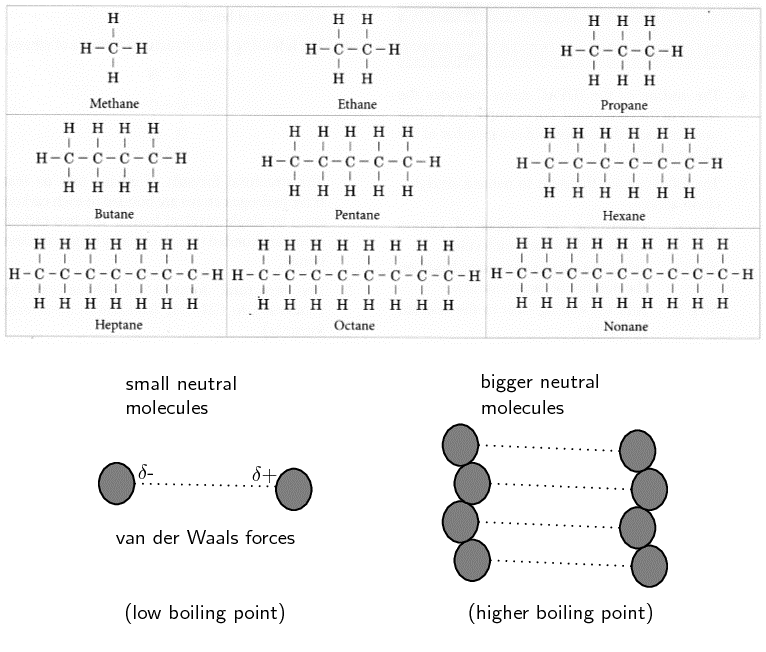
Remember that the alkanes are a group of organic compounds that contain carbon and hydrogen atoms bonded together. The carbon atoms link together to form chains of varying lengths. Remember that the alkanes are relatively unreactive because of their stable [latex]\scriptsize \displaystyle \text{C-C}[/latex] and [latex]\scriptsize \displaystyle \text{C-H}[/latex] bonds. The boiling points and melting points of these molecules are determined by their molecular structure and their surface area.
The more carbon atoms there are in an alkane, the greater the surface area available for intermolecular interactions.
| Formula | Name | Molecular mass ([latex]\scriptsize \text{g}\text{.mo}{{\text{l}}^{{\text{-1}}}}[/latex]) |
Melting point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) | Boiling point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) | Phase (at [latex]\scriptsize 25\text{ }{}^\circ \text{C}[/latex]) |
| [latex]\scriptsize \displaystyle \text{C}{{\text{H}}_{\text{4}}}[/latex] | methane | [latex]\scriptsize 16.04[/latex] | [latex]\scriptsize \displaystyle -182[/latex] | [latex]\scriptsize \displaystyle -162[/latex] | gas |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}[/latex] | ethane | [latex]\scriptsize \displaystyle 30.06[/latex] | [latex]\scriptsize \displaystyle -183[/latex] | [latex]\scriptsize \displaystyle -89[/latex] | gas |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{3}}}{{\text{H}}_{\text{8}}}[/latex] | propane | [latex]\scriptsize \displaystyle 44.08[/latex] | [latex]\scriptsize -188[/latex] | [latex]\scriptsize -42[/latex] | gas |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{4}}}{{\text{H}}_{{\text{10}}}}[/latex] | butane | [latex]\scriptsize 58.1[/latex] | [latex]\scriptsize -137[/latex] | [latex]\scriptsize 0[/latex] | gas |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{6}}}{{\text{H}}_{{\text{14}}}}[/latex] | hexane | [latex]\scriptsize 86.14[/latex] | [latex]\scriptsize -95[/latex] | [latex]\scriptsize 68.5[/latex] | liquid |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{8}}}{{\text{H}}_{{\text{18}}}}[/latex] | octane | [latex]\scriptsize 114.18[/latex] | [latex]\scriptsize -57[/latex] | [latex]\scriptsize 125.5[/latex] | liquid |
| [latex]\scriptsize \displaystyle {{\text{C}}_{{\text{20}}}}{{\text{H}}_{{\text{42}}}}[/latex] | icosane | [latex]\scriptsize 282.42[/latex] | [latex]\scriptsize 37[/latex] | [latex]\scriptsize 343[/latex] | solid |
Notice that when the molecular mass of the alkanes is low (i.e. there are few carbon atoms), the organic compounds are gases because the intermolecular forces are weak. As the number of carbon atoms and the molecular mass increases, the compounds are more likely to be liquids or solids because the intermolecular forces are stronger.
The larger a molecule is, the stronger the intermolecular forces are between the molecules. This is one of the reasons why methane is a gas at room temperature while hexane is a liquid and icosane is a solid.
Be careful when comparing molecules with different types of intermolecular forces. For example, small molecules with hydrogen bonding can have stronger intermolecular forces than large molecules with only van der Waals forces.
also increases with molecular size. Flash point increases with increasing chain length meaning that the longer chains are less flammable although they can still be ignited.
It is partly the stronger intermolecular forces that explain why petrol (mainly octane) is a liquid, while candle wax ([latex]\scriptsize \displaystyle {{\text{C}}_{{\text{23}}}}{{\text{H}}_{{\text{48}}}}[/latex]) is a solid. If these intermolecular forces did not increase with increasing molecular size, we would not be able to put liquid fuel into our cars or use solid candles.
Exercise 3.2
Refer to the data table below which shows the melting point and boiling point for several organic compounds with different functional groups.
| Formula | Name | Melting point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) | Boiling point ([latex]\scriptsize \text{ }\!\!{}^\circ\!\!\text{ C}[/latex]) |
| [latex]\scriptsize {{\text{C}}_{\text{4}}}{{\text{H}}_{{\text{10}}}}[/latex] | Butane | [latex]\scriptsize -137[/latex] | [latex]\scriptsize 0[/latex] |
| [latex]\scriptsize {{\text{C}}_{5}}{{\text{H}}_{{\text{12}}}}[/latex] | Pentane | [latex]\scriptsize -130[/latex] | [latex]\scriptsize 36[/latex] |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{6}}}{{\text{H}}_{{\text{14}}}}[/latex] | Hexane | [latex]\scriptsize -95[/latex] | [latex]\scriptsize 68.5[/latex] |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}[/latex] | But-1-ene | [latex]\scriptsize -185[/latex] | [latex]\scriptsize -6.5[/latex] |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{10}}}}[/latex] | Pent-1-ene | [latex]\scriptsize -165.2[/latex] | [latex]\scriptsize 30[/latex] |
| [latex]\scriptsize \displaystyle {{\text{C}}_{\text{6}}}{{\text{H}}_{{\text{12}}}}[/latex] | Hex-1-ene | [latex]\scriptsize -140[/latex] | [latex]\scriptsize 63[/latex] |
- At room temperature (approx. [latex]\scriptsize 25\text{ }{}^\circ \text{C}[/latex]. ), which of the organic compounds in the table are gases?
- In the alkanes, describe what happens to the melting point and boiling point as the number of carbon atoms in the compound increases. Explain your answer.
The full solutions are at the end of the unit.
Chemical properties
The term ‘chemical properties’ describes the way that an element or compound reacts chemically with other substances. A chemical property is a characteristic or behaviour of a substance that may be observed when it undergoes a chemical change or reaction.
Chemical properties are seen either during or following a reaction since the arrangement of atoms within a sample must be disrupted for the property to be investigated. This is different from a physical property, which is a characteristic that may be observed and measured without changing the chemical identity of a specimen.
Examples of chemical properties include flammability, toxicity, chemical stability, and heat of combustion.
The chemical properties of the alkanes
Because there are only [latex]\scriptsize \displaystyle \text{C-H}[/latex] bonds throughout the whole structure of alkanes and these bonds are quite stable, and do not want to break easily, the alkanes are quite inert.
Alkanes undergo only two reactions:
- Combustion reaction with oxygen: alkane + oxygen ➝ carbon dioxide + water
.
For example: [latex]\scriptsize \displaystyle \text{C}{{\text{H}}_{\text{4}}}_{{\left( \text{g} \right)}}\text{+ 2}{{\text{O}}_{\text{2}}}_{{\left( \text{g} \right)}}\to \text{C}{{\text{O}}_{\text{2}}}_{{\left( \text{g} \right)}}\text{+2}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{l} \right)}}}[/latex] - Substitution reaction with halogens: alkane + halogen ➝ halogenoalkane + hydrogen halide
.
These types of reactions need sunlight or a UV light. For example:
.
[latex]\scriptsize \displaystyle \text{C}{{\text{H}}_{\text{4}}}_{{\left( \text{g} \right)}}\text{+B}{{\text{r}}_{\text{2}}}_{{\left( \text{g} \right)}}\to \text{C}{{\text{H}}_{\text{3}}}\text{B}{{\text{r}}_{{\left( \text{g} \right)}}}\text{+HB}{{\text{r}}_{{\left( \text{g} \right)}}}[/latex]
The chemical properties of alkenes
In alkenes the functional group is a carbon-to-carbon double bond ([latex]\scriptsize \displaystyle \text{C=C}[/latex]). This bond is quite unstable and breaks quite easily. This makes alkenes more reactive than alkanes. Unlike alkanes, alkenes undergo many reactions.
- Combustion with oxygen: alkenes also react with oxygen in the air when it is ignited to produce carbon dioxide and water. But unlike alkanes they do not burn so cleanly.
.
For example: [latex]\scriptsize \displaystyle {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}_{{\left( \text{g} \right)}}\text{+}{{\text{O}}_{\text{2}}}_{{\left( \text{g} \right)}}\to \text{2C}{{\text{O}}_{\text{2}}}_{{\left( \text{g} \right)}}\text{+2}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{l} \right)}}}[/latex] - Addition reaction with halogens: the reaction normally takes place at room temperature and pressure. The reaction is called ‘addition’ because the two halogen atoms are being added to the hydrocarbon.
.
For example: ethene + bromine ➝ 1,2-dibromoethane
.
[latex]\scriptsize \displaystyle {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}_{{\left( \text{g} \right)}}\text{+B}{{\text{r}}_{\text{2}}}_{{\left( \text{l} \right)}}\to {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}\text{B}{{\text{r}}_{\text{2}}}_{{\left( \text{l} \right)}}[/latex] - Hydration: In the hydration reaction, ethene is reacted with steam to produce ethanol. The ethanol which is produced by the hydration process is extremely pure. Ethanol is produced in this way in factories.
.
For example: ethene + steam ➝ ethanol
.
[latex]\scriptsize \displaystyle {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}_{{\left( \text{g} \right)}}\text{+ }{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{g} \right)}}}\to {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}\text{O}{{\text{H}}_{{\left( \text{g} \right)}}}[/latex]
The chemical properties of alcohols
The OH bond is quite polar which makes the alcohols quite reactive, so the alcohol undergoes several chemical reactions.
- Reaction with oxygen: When ignited, alcohols burn or react with oxygen to produce carbon dioxide and water. Alcohol is a very good fuel. Alcohol produced by fermenting sugar cane (or other plant material) is used as a fuel is known as biofuel.
.
For example: ethanol + oxygen ➝ carbon dioxide + water
.
[latex]\scriptsize \displaystyle {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}\text{O}{{\text{H}}_{{\left( \text{l} \right)}}}\text{+3}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\to \text{2C}{{\text{O}}_{{\text{2}\left( \text{g} \right)}}}\text{+3}{{\text{H}}_{\text{2}}}{{\text{O}}_{{\left( \text{l} \right)}}}[/latex] - Dehydration: Dehydration means the loss of water. In this reaction the alcohol loses a water molecule and forms an alkene of the same carbon number. So if ethanol undergoes the dehydration reaction, the corresponding alkene produced will be ethene.
The chemical properties of haloalkanes
Haloalkanes are quite reactive compounds. They undergo several different types of reactions. They also react with some metals to form organometallic compounds. The reactivity of haloalkanes is mainly due to the polar nature of the carbon-halogen bond.
Summary
In this unit you have learnt the following:
- The melting and boiling points of alkanes, alkenes and alkynes increase as the carbon number increases.
- Alkanes have relatively higher melting and boiling points than alkenes. This is because the presence of carbon-to-carbon double bond in alkenes reduces the effectiveness of the intermolecular forces between the molecules.
- Alkanes and alkenes are insoluble in water. This is because their weak London forces cannot disrupt the much stronger hydrogen bonds between the water molecules.
- Alcohols have much higher melting and boiling points compared to alkanes and alkenes as they can form hydrogen bonds.
- Alcohols are highly soluble in water as they can form hydrogen bonding with the water molecules.
- Alkanes do not have a functional group and undergo two chemical reactions: combustion and substitution.
- In alkenes the functional group is the carbon-to-carbon double bond. Alkenes undergo three chemical reactions: combustion, addition and hydration.
- In alcohols, the functional group is the OH group. Alcohols undergo two chemical reactions: combustion and dehydration.
Unit 3: Assessment
Suggested time to complete: 15 minutes
- Which container (A or B) contains the compound with higher vapour pressure in it? Explain your answer.

- Refer to the data table below which shows the melting points and boiling points for several organic compounds with different functional groups.
Formula Name Melting point (℃) Boiling point (℃) [latex]\scriptsize {{\text{C}}_{\text{4}}}{{\text{H}}_{{\text{10}}}}[/latex] butane [latex]\scriptsize -137[/latex] [latex]\scriptsize 0[/latex] [latex]\scriptsize {{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}[/latex] but-1-ene [latex]\scriptsize -185[/latex] [latex]\scriptsize -6.5[/latex] [latex]\scriptsize {{\text{C}}_{\text{4}}}{{\text{H}}_{{\text{10}}}}\text{O}[/latex] butan-1-ol [latex]\scriptsize -90[/latex] [latex]\scriptsize 118[/latex] [latex]\scriptsize {{\text{C}}_{5}}{{\text{H}}_{{\text{12}}}}[/latex] pentane [latex]\scriptsize -130[/latex] [latex]\scriptsize 36[/latex] [latex]\scriptsize {{\text{C}}_{5}}{{\text{H}}_{{\text{10}}}}[/latex] pent-1-ene [latex]\scriptsize -165.2[/latex] [latex]\scriptsize 30[/latex] [latex]\scriptsize {{\text{C}}_{5}}{{\text{H}}_{{\text{12}}}}\text{O}[/latex] pentan-1-ol [latex]\scriptsize -78[/latex] [latex]\scriptsize 138[/latex] - At room temperature (approx. [latex]\scriptsize \displaystyle 25\text{ }{}^\circ \text{C}[/latex]), which of the organic compounds in the table are:
- gases
- liquids
- Look at the alkane, alkene, and an alcohol with 4 carbon atoms:
- How do their melting and boiling points compare?
- Explain why their melting points and boiling points are different?
- At room temperature (approx. [latex]\scriptsize \displaystyle 25\text{ }{}^\circ \text{C}[/latex]), which of the organic compounds in the table are:
- The following diagram shows the reactants in an addition reaction:
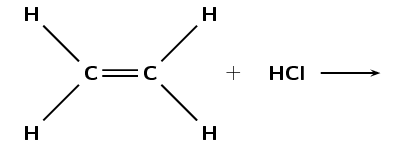
- Draw the structural representation of the final product in this reaction.
- What is the condensed structural representation of the product?
- Which types of homologous series can undergo additional reactions? Is this a saturated or unsaturated series?
The full solutions are at the end of the unit.
Unit 3: Solutions
Exercise 3.1
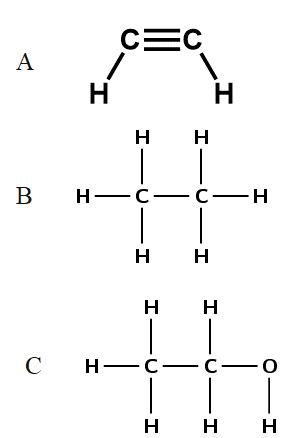
- ethane and ethyne
- .
A – induced dipole
.
B – hydrogen bonding
.
C – hydrogen bonding - B has only induced-dipole forces (weak van der Waals forces). C has hydrogen bonding. Since hydrogen bonding is a stronger intermolecular force than van der Waals forces, more energy is required to separate the molecules of ethanol than the molecules of ethane. Thus ethanol has a higher boiling point than ethane.
- .
Exercise 3.2
- 1. butane and butene
- The melting and boiling points increase as the number of carbon atoms increase. As the number of carbon atoms increases, so does the surface area of the molecule. This leads to stronger intermolecular forces, which are responsible for the increase in melting and boiling points.
Unit 3: Assessment
- B. There are more molecules of B in the vapour phase than there are of A. More molecules in the vapour phase means a higher vapour pressure.
- .
- .
- butane, but-1-ene
- all except butane and but-1-ene
- .
- highest boiling (and melting) point is butan-1-ol, then butane (the alkane) followed by but-1-ene (the alkene).
- The alcohols can only form strong hydrogen bonds between molecules leading to higher melting and boiling points. The alkenes and alkanes form weak induced-dipole forces. However, the alkenes are unsaturated and so the melting and boiling points of an alkene are lower than those of an alkane with the same number of carbon atoms.
- .
- .
- .
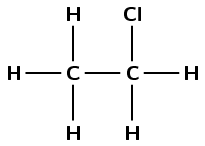
- [latex]\scriptsize \displaystyle \text{C}{{\text{H}}_{\text{3}}}\text{C}{{\text{H}}_{\text{2}}}\left( {\text{Cl}} \right)[/latex]
- .
- Any unsaturated series where the functional group contains double or triple bonds (e.g. alkenes, alkynes).
Media Attributions
- Fig 1 © Siyavula is licensed under a CC BY-NC (Attribution NonCommercial) license
- Fig 2 © DHET is licensed under a CC BY (Attribution) license
- Fig 3 © Siyavula is licensed under a CC BY-NC (Attribution NonCommercial) license
- Fig 4 © DHET is licensed under a CC BY (Attribution) license
- Fig 5 © Siyavula is licensed under a CC BY-NC (Attribution NonCommercial) license
- Fig 6 © Siyavula is licensed under a CC BY-NC (Attribution NonCommercial) license
- Fig 7 © DHET is licensed under a CC BY (Attribution) license
- Fig 8 © Siyavula is licensed under a CC BY-NC (Attribution NonCommercial) license
the pressure exerted (at a specific temperature) on a solid or liquid compound by molecules of that compound that are in the gas phase
the tendency of molecules at the surface of a compound to enter the gas phase
a measure of the mass per unit of volume
